Understanding the preferences of clinicians, patients, and payers
Discrete Choice Experiment (DCE)
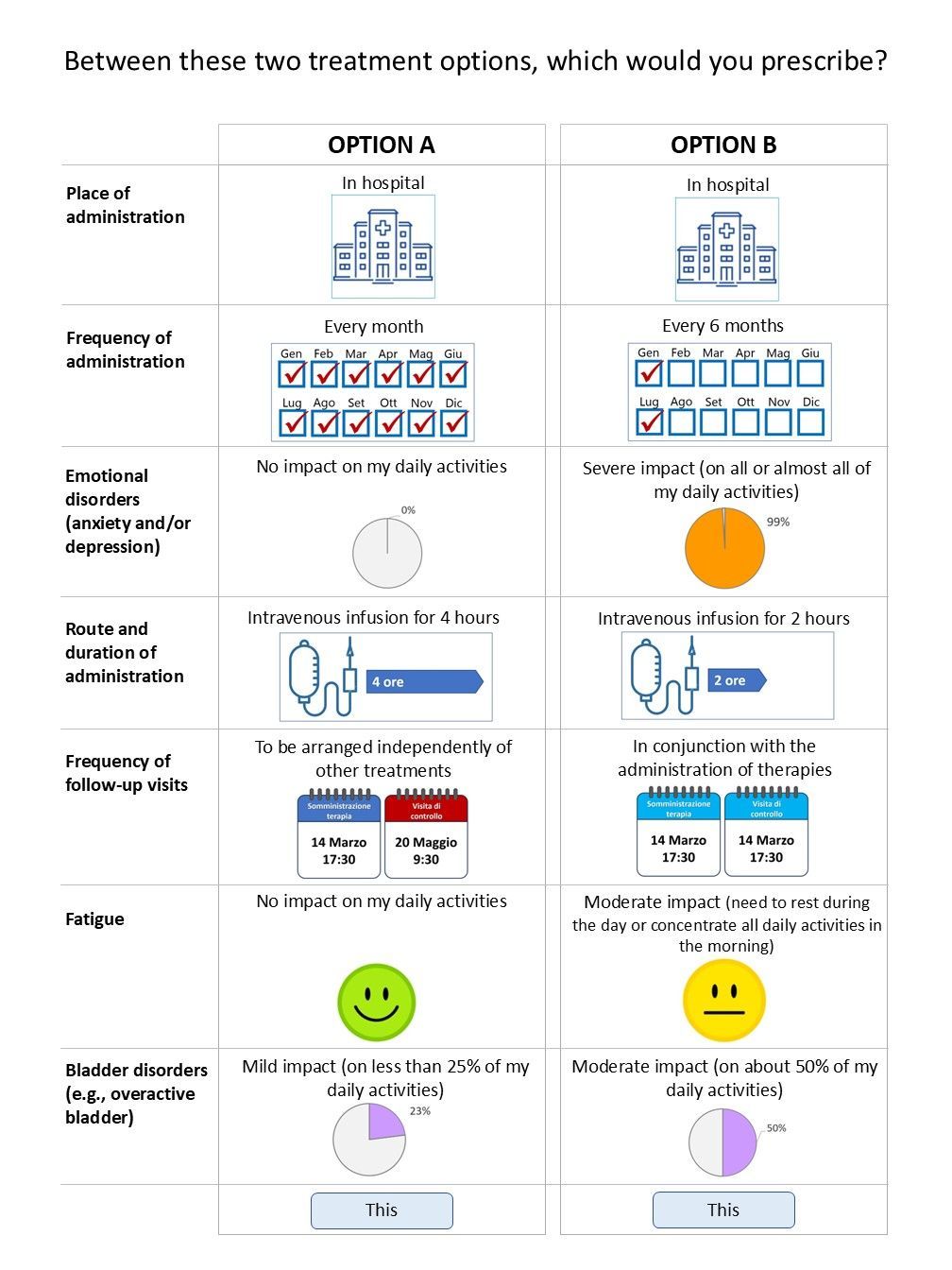
Understanding preferences
In recent years, there has been growing interest among the scientific community in methodologies that allow researchers to better understand the preferences of clinicians and patients with regard to various issues in the healthcare and pharmaceutical sectors.
This is confirmed by the steady increase (around 20% each year) in publications using these methodologies, particularly the Discrete Choice Experiment.
Calibra's goal is to rigorously evaluate the preferences of patients, clinicians, and payors in order to incorporate them into the development, introduction, and consolidation of new drugs and new therapeutic approaches in clinical practice.
To quantify preferences, DCE participants are asked to compare scenarios that are constructed hypothetically and randomly.
The value of preferences
The results of DCE studies have a significant impact on treatment adherence because physicians can make choices that take patient preferences into account. The scientific literature clearly shows that when a treatment aligns with patient preferences, adherence to therapy improves, leading to better clinical outcomes. This is therefore a result that meets the expectations of the patient, the clinician, and the health authorities.
Publication of results
All DCE projects developed by Calibra have been published in indexed scientific journals.
Strengths of the Discrete Choice Experiment
- Stakeholders: allows you to involve clinicians, patients, payors, and nurses, as desired
- Patient associations: work with them by involving the patient
- Value of the results:
- Useful for Marketing, Market Access, Medical Dept
- The data is quantitative in nature, relating to the preferences attributed by various stakeholders to certain characteristics of therapies (route of administration, frequency of administration, efficacy, side effects, place of administration) and/or access pathways
- The preferences expressed by different stakeholders can be explored in depth from a pharmacoeconomic perspective, providing data that can be used in the registration and negotiation process with health authorities
- DCE studies are published in indexed international journals
- Studies on preferences are increasingly important for health authorities (Project PREFER)
- Timeframe: results can be obtained in 6-8 months
- Costs: attractive in terms of the quality and usefulness of the data obtained and the timeframe
Possible applications of DCE

When to apply the DCE

Patients
Patients' preferences can be expressed in terms of the importance of what matters most to them in relation to treatments (route of administration, frequency of administration, efficacy, side effects, place where treatment is administered), expressing how much risk they consider acceptable in order to obtain a certain benefit (trade-off) as well as the compromises they are willing to make between the characteristics of different treatments or, more generally, healthcare interventions.
Clinicians
In addition to patients, the Discrete Choice Experiment is also normally conducted with clinicians to understand their preferences regarding therapies and access pathways and compare them with those of patients.
Payors
The Discrete Choice Experiment also applies to payors who must express their opinion on the value of innovation in new therapies and the cost to be incurred for the reimbursement of a new treatment.
For example, in the case of a highly innovative but very expensive oncology drug, it is important to know the criteria that guide the choices of the payer called upon to make a decision, i.e., to establish the trade-off between the value and improvement offered by the new drug and the cost of introducing this innovation.
The value of preferences for international health authorities (FDA, EMA) and Italian health authorities (AIFA)
In recent years, there has been a significant increase in the number of scientific publications based on Discrete Choice Experiments in the medical and clinical fields. In particular, various health authorities such as the FDA, EMA, and AIFA have expressed interest and initiated studies (e.g., the PREFER Project) on the use and evaluation of patient and clinician preferences in drug registration decision-making processes.
The increasingly important role of the patient and the growing focus on the issue of therapeutic adherence and patient quality of life have convinced these institutions to identify tools that can make a significant contribution in this direction, evaluating the opportunity to make surveys of this type fundamental, if not binding, for obtaining drug registration.
The PREFER Project
Over the past five years, the European Medicines Agency (EMA) supported the PREFER Project, published in 2024, which highlights the importance of evaluating the preferences and choice algorithms of patients and clinicians with a view to improving treatment adherence.
The FDA and the EMA have published several articles on the growing importance of preferences in regulatory processes (e.g., How Patient Preferences Contribute to Regulatory Decisions for Medical Devices, September 25, 2017, by the FDA; Patient Preference Information (PPI) in Medical Device Decision Making, July 3, 2023 by FDA; Qualification Opinion of IMI PREFER, May 3, 2022 EMADOC-1700519818-808373 Committee for Medicinal Products for Human Use).
Preferences for the registration dossier for AIFA
The results of DCE studies can be attached to registration dossiers to support the approval of a new drug, new therapeutic indications, or new formulations.
Organizational characteristics of DCE projects
DCE projects are coordinated by a Scientific Board.
Clinicians, patients, payors, nurses, and pharmacists can participate depending on the objectives and issues that the project aims to address.
Sample: the number of participants depends on the characteristics of the project and can be calculated precisely to obtain reliable data.
In general, there may be between 40 and 100 or more participating physicians, depending on the disease and the multidisciplinary nature of the treatment.
There may be between 50-60 and 500-1000 patients. They are usually involved through patient associations in the therapeutic area of interest.
Timeframe: 6-8 months from project kick-off to submission of the paper for publication.
IT platform for DCE and other projects
For its statistical and data analysis activities, Calibra uses the application developed by the American company Sawtooth Software, the platform most widely used by the scientific community.